New Biological therapeutics to control hemolysis-driven disease
We are establishing a bench-to-application translational platform for the development of haptoglobin and hemopexin as therapeutics in hemolytic diseases. Ten years ago, we discovered that the plasma protein haptoglobin mitigates hemodynamic and pro-oxidative adverse effects that emerge from cell-free hemoglobin following hemolysis. Based on these initial observations, we have developed a comprehensive biochemical and physiological model of cell-free hemoglobin and heme toxicity that applies to multiple disease conditions such as rare hemolytic anemias (e.g., sickle cell disease), blood transfusion, sepsis, and device-induced hemolysis. In all these conditions cell-free hemoglobin toxicity amplifies vascular, renal, and inflammatory disease processes leading to adverse outcomes. Our ultimate aim is the development of a biological scavenger protein drug that blocks hemoglobin toxicity in these patients.
Hemolysis associated pathologies.
(adapted from Buehler PW et al., 2020 and Schaer CA et al., 2015).
READINGS
Modular Platform for the Development of Recombinant Hemoglobin Scavenger Biotherapeutics
Buzzi RM, Owczarek CM, Akeret K, Tester A, Pereira N, Butcher R, Brügger-Verdon V, Hardy MP, Illi M, Wassmer A, Vallelian F, Humar R, Hugelshofer M, Buehler PW, Gentinetta T, Schaer DJ. (2021).
Molecular Pharmaceutics.
Abstract
Cell-free hemoglobin (Hb) is a driver of disease progression in conditions with intravascular or localized hemolysis. Genetic and acquired anemias or emergency medical conditions such as aneurysmal subarachnoid hemorrhage involve tissue Hb exposure. Haptoglobin (Hp) captures Hb in an irreversible protein complex and prevents its pathophysiological contributions to vascular nitric oxide depletion and tissue oxidation. Preclinical proof-of-concept studies suggest that human plasma-derived Hp is a promising therapeutic candidate for several Hb-driven diseases. Optimizing the efficacy and safety of Hb-targeting biotherapeutics may require structural and functional modifications for specific indications. Improved Hp variants could be designed to achieve the desired tissue distribution, metabolism, and elimination to target hemolytic disease states effectively. However, it is critical to ensure that these modifications maintain the function of Hp. Using transient mammalian gene expression of Hp combined with co-transfection of the pro-haptoglobin processing protease C1r-LP, we established a platform for generating recombinant Hp-variants. We designed an Hpβ-scaffold, which was expressed in this system at high levels as a monomeric unit (mini-Hp) while maintaining the key protective functions of Hp. We then used this Hpβ-scaffold as the basis to develop an initial proof-of-concept Hp fusion protein using human serum albumin as the fusion partner. Next, a hemopexin-Hp fusion protein with bispecific heme and Hb detoxification capacity was generated. Further, we developed a Hb scavenger devoid of CD163 scavenger receptor binding. The functions of these proteins were then characterized for Hb and heme-binding, binding of the Hp-Hb complexes with the clearance receptor CD163, antioxidant properties, and vascular nitric oxide sparing capacity. Our platform is designed to support the generation of innovative Hb scavenger biotherapeutics with novel modes of action and potentially improved formulation characteristics, function, and pharmacokinetics.
Haptoglobin Therapeutics and Compartmentalization of Cell-Free Hemoglobin Toxicity
Buehler PW, Humar R, Schaer DJ. (2020).
Trends in Molecular Medicine.
Abstract
Haptoglobin Preserves Vascular Nitric Oxide Signaling during Hemolysis
Christian A. Schaer, Jeremy W. Deuel, Daniela Schildknecht, Leila Mahmoudi, Ines Garcia-Rubio, Catherine Owczarek, Stefan Schauer, Reinhard Kissner, Uddyalok Banerjee, Andre F. Palmer, Donat R. Spahn, David C. Irwin, Florence Vallelian, Paul W. Buehler, Dominik J. Schaer. (2015).
American journal of respiratory and critical care medicine, 193(10), 1111-1122.
Abstract
Hemolysis occurs not only in conditions such as sickle cell disease and malaria but also during transfusion of stored blood, extracorporeal circulation, and sepsis. Cell-free Hb depletes nitric oxide (NO) in the vasculature, causing vasoconstriction and eventually cardiovascular complications. We hypothesize that Hb-binding proteins may preserve vascular NO signaling during hemolysis. Objectives:
Characterization of an archetypical function by which Hb scavenger proteins could preserve NO signaling during hemolysis.
Methods:
We investigated NO reaction kinetics, effects on arterial NO signaling, and tissue distribution of cell-free Hb and its scavenger protein complexes.
Measurements and Main Results:
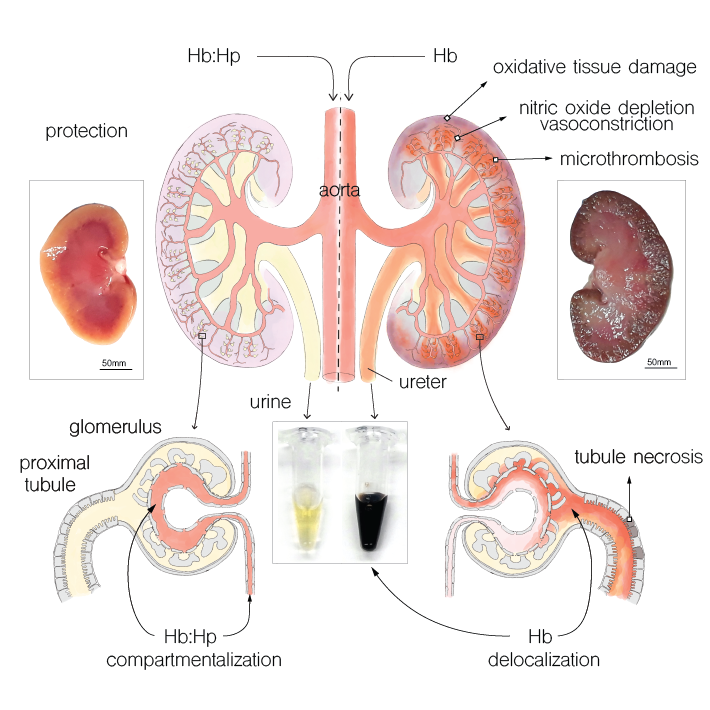
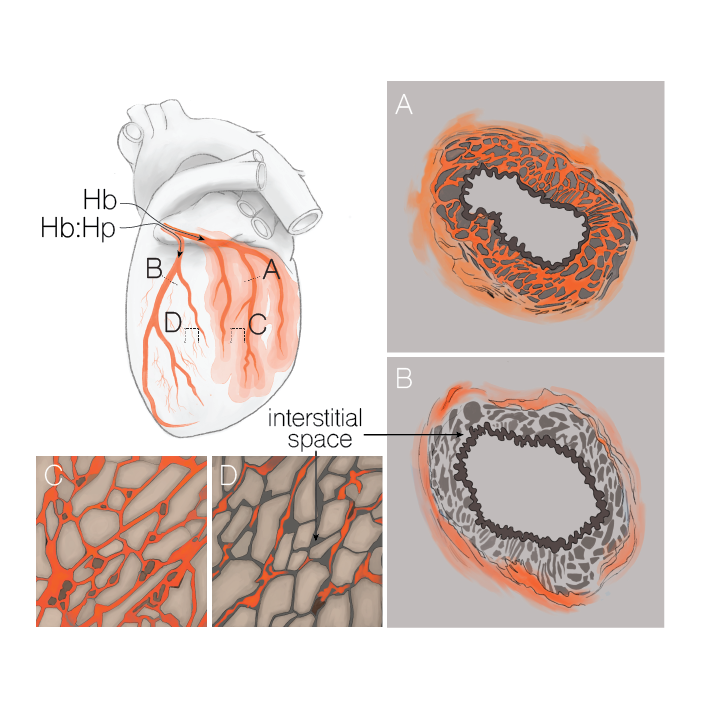
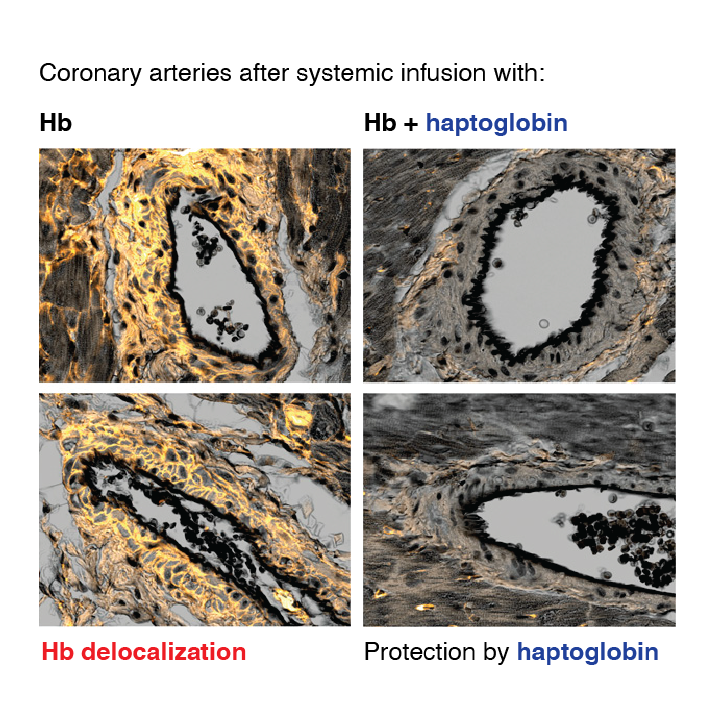
Extravascular translocation of cell-free Hb into interstitial spaces, including the vascular smooth muscle cell layer of rat and pig coronary arteries, promotes vascular NO resistance. This critical disease process is blocked by haptoglobin. Haptoglobin does not change NO dioxygenation rates of Hb; rather, the large size of the Hb:haptoglobin complex prevents Hb extravasation, which uncouples NO/Hb interaction and vasoconstriction. Size-selective compartmentalization of Hb functions as a substitute for red blood cells after hemolysis and preserves NO signaling in the vasculature. We found that evolutionarily and structurally unrelated Hb-binding proteins, such as PIT54 found in avian species, functionally converged with haptoglobin to protect NO signaling by sequestering cell-free Hb in large protein complexes.
Conclusions:
Sequential compartmentalization of Hb by erythrocytes and scavenger protein complexes is an archetypical mechanism, which may have supported coevolution of hemolysis and normal vascular function. Therapeutic supplementation of Hb scavengers may restore vascular NO signaling and attenuate disease complications in patients with hemolysis.
Sequestration of extracellular hemoglobin within a haptoglobin complex decreases its hypertensive and oxidative effects in dogs and guinea pigs
Boretti FS, Buehler PW, D’Agnillo F, Kluge K, Glaus T, Butt OI, Jia Y, Goede J, Pereira CP, Maggiorini M, Schoedon G, Alayash AI, Schaer DJ. (2009).
The Journal of clinical investigation, 119(8), 2271-2280.
https://doi.org/10.1172/JCI39115
Abstract
Release of hemoglobin (Hb) into the circulation is a central pathophysiologic event that contributes to morbidity and mortality in chronic hemolytic anemias and severe malaria. These toxicities arise from Hb-mediated vasoactivity, possibly due to NO scavenging and localized tissue oxidative processes. Currently, there is no established treatment that targets circulating extracellular Hb. Here, we assessed the role of haptoglobin (Hp), the primary scavenger of Hb in the circulation, in limiting the toxicity of cell-free Hb infusion. Using a canine model, we found that glucocorticoid stimulation of endogenous Hp synthesis prevented Hb-induced hemodynamic responses. Furthermore, guinea pigs administered exogenous Hp displayed decreased Hb-induced hypertension and oxidative toxicity to extravascular environments, such as the proximal tubules of the kidney. The ability of Hp to both attenuate hypertensive responses during Hb exposure and prevent peroxidative toxicity in extravascular compartments was dependent on Hb-Hp complex formation, which likely acts through sequestration of Hb rather than modulation of its NO- and O2-binding characteristics. Our data therefore suggest that therapies involving supplementation of endogenous Hb scavengers may be able to treat complications of acute and chronic hemolysis, as well as counter the adverse effects associated with Hb-based oxygen therapeutics.